Table of Contents
2. Anatomy of the Eye and Orbit (Mission for Vision)
3. Anatomy of the Eye, Orbit with Neuroanatomy (Medrounds)
4. Ocular Pathology Tutorial (Mission for Vision)
5. Processing Manual for Eye Pathology (Medrounds)
6. Self Test and Board Review for Ophthalmology (Medrounds)
7. Test for Ocular Cytopathology- the Atlas
Methods in Histology
1. Surgical Removal of Tissue- In the laboratory, tissue is usually obtained from patients undergoing surgery. The tissue is analysed to make diagnoses as to the cause or histologic manifestion of the patient's problem. In each case, particular questions may be asked such as "Is it cancer?". Tissues for research are obtained in a variety of ways with approval from the Institution Review Board (IRB) and patient's consent if needed. In any case the preparation of tissue from the operating room is critical. For most histologic work, tissue will require preservation which may call for freezing or chemical "fixation".
2. Fixation- Fixation is the interruption of dynamic cell processes such as self destruction from enzymes. There are many methods of fixation but the common thread is to render the structural elements insoluble via chemicals that precipitate proteins and other compounds. Various fixatives yield different visual and biochemical results and sometimes special fixatives are desired. The vast majority of tissue will be received in some chemical fixative from the operating room. If consulted ahead of time the pathology laboratory selects the type of fixative. Here are the most commonly used fixatives in eye pathology:- Formalin- Neutral buffered formalin (10%) produces very little nuclear clumping and works by crosslinking proteins and DNA. Formalin penetrates tissue at a rate of about 1 mm /hour. Proteins are crosslinked via methylene bridges between amino groups. DNA can be crosslinked with proteins. Specifically Lys, Cys, His, and Trp cross link with dG, dA, and dC. Similarly DNA is crosslinked and denatured with interruption of interchain hydrogen bonds in AT-rich regions. DNA isolated in fomalin fixed tissue has variable sequence alterations that hinder its use for accurate sequencing. In general formalin is excellent for preserving most antigens for antibody reactions (used in immunohistochemical reactions).
- Ethanol- 50-80% acidified ethanol is an excellent fixative that appears to precipitate proteins and nuclei acids with little cross linking. This leaves DNA and even RNA intact for analysis. DNA and RNA are often stored for long periods in ethanol. Acidified ethanol is much better for preservation of DNA and RNA than formalin. For genetic studies ethanol is an excellent fixative to prepare the samples when DNA sequencing may be needed, such as in a tumor of children called retinoblastoma. Immunohistochemistry (intact antigens) may work very well in alcohol fixation but this is not guaranteed because as with formalin, epitopes may be altered by fixation. However, the disadvantage of alcohol is that it causes clumping of nuclear chromatin and shrinkage of cells that gives the appearance of nuclear atypia under the microscope. Actually the normal cells may appear atypical to the untrained eye because the nuclear clumping and shrinkage mimics the appearance of cancer cells. However, there are numerous advantages. The acidified ethanol mixture may be just a combination of vinegar and ethanol and can be relatively non-toxic, an asset for specimens referred from a far distance and transported through the a mail service. Formalin is poisonous. Also, alcohol and vinegar are household substances that are readily accessible to clients that don't keep formalin in stock.
- Paraformaldehyde- A polymer of 8-100 formaldehyde units, paraformaldehyde has the advantage of adequate fixation for electron microscopy and presumably somewhat reversible. Although expensive to maintain in the laboratory (out of light and refrigerated) paraformaldehyde is an excellent fixative when one is uncertain of the pathology. DNA extraction from paraformadehyde is somewhat similar to formalin and not ideal. Although presumably reversible, cross linking of DNA does seem to occur that limits the yield.
- Freezing- Freezing is an excellent preservative that keep antigens, DNA, and RNA relatively intact. For this purpose, rapid freezing is a preferred method of preservation for diseases in which the antibody epitopes are destroyed by cross linking. Rapid freezing also is excellent (like ethanol) when genetic studies are needed, particularly if RNA is required. Although acidified ethanol works, dry ice or liquid nitrogen may be the method of choice if readily available. The disadvantage is that the tissue must be kept frozen and shipped and stored at low temperature. This is expensive.
- Preservative Buffer- Tissue may be kept in some sterile neutral buffer (such as phosphate buffered saline) for several hours especially if kept cold. Specimens in the buffer can be transferred to the desired fixative or frozen once they reach the laboratory. This is a preferred way to handle ocular cicatricial pemphigoid cases that are sent locally where shipping can be occur in the same day. Buffers are used for specimens for ocular cicatricial pemphigoid, a disease where fluorescent antibodies are directly applied to sections in order to visualize basement membrane antigens. These antigens are not stable with fixatives such as formalin, paraformaldehyde and perhaps ethanol. So called Zeus Fixative provides stability for 1 day and is composed of potassium citrate, n-ethyl maleimide, magnesium sulfate and ammonium sulfate. Zeus Media is used for transport for outside specimens to a referral laboratory. It is not ideal.
- Osmication- Lipids are incidentally extracted from tissue by organic solvents used in histologic processing for paraffin embedding. Lipid removal can be vitiated by fixation with osmium tetroxide. Osmication is usually done in the electron microscopy laboratory. Osmium tetroxide is used to fix the lipids of a cancer of the eyelids known as sebaceous cell carcinoma. The lipids can also be preserved by freezing, because no organic solvents are used in processing this tissue prior to microtomy. Specimens fixed in this manner are generally embedded in a plastic resin and very fine 1 micron "thick" sections are made with a special microtome. The plastic sections (called "thick" sections) can be stained with toluidine blue, a dye that stains lipid green and viewed with a light microscope. If needed ultrathin sections (nm thick) can be made for electron microscopy.
- Decalcification- Bone fragments are very hard due to the presence of calcified osteoid and can ruin the microtome blade if not softened prior to sectioning. Calcium is removed by a process called decalcification, which uses acid and/or EDTA for removal. It is important that the specimen be well fixed prior to decalcification. Many of the decalcification solutions contain formic acid, hydrochloric acid and acetic acid. These are compatible with different fixatives. Formic acid works well with formalin. It is important to wash the specimen after decalcification with water and then to place it in ethanol. The acidic solutions are also inherently fixative although when used alone in high concentration can deleteriously affect the morphologic appearance.
3. Histologic Processing, Sectioning and Staining.- Tissues that have been fixed need to be infiltrated with some solution of paraffin, gelatin, celloidin, or plastic that later hardens. Tissue can then be cut into very thin sections. Paraffin is the most common infiltration and embedding compound and requires that the water be removed (dehydration). This is accomplished by using progressively more concentrated alcohols, e.g 50, 70, 80, 95, 100% with several changes at 95, and 100% and then an organic solvent like xylene for several changes with final immersion into a hot paraffin bath. The tissue can then be carefully mounted in a slab of paraffin (called a paraffin block) for fine cutting (microtomy). Histologic sections cut from a paraffin block with a rotary microtome are generally 4-6 microns in thickness in experienced hands. This is a very important skill that some lab members will need to know well. Histologic stains are varied and can be used for visualization of specific features. In general, the laboratory uses hematoxylin and eosin stains (H&E) for almost every tissue as a start. Nuclei are stained blue and cytoplasm pink with H&E. H&E is a work horse stain that is good for a general view. Periodic acid-Schiff (PAS) is an excellent stain for carbohydrates. For example mucins in the conjunctiva and cornea stain bright pink with this stain. There are a litany of other special histologic stains used by the laboratory and varied antibodies available for immunohistochemistry.
4. Microscopic Analysis- The most common microscope used in the laboratory is the compound light microscope. Visible light shines through the stained tissue section and is collected by objectives and condensing lenses. The resolution of the microscope is limited by the numerical aperature of the lenses and approaches about .25 microns. For reference a red blood cell is about 7-10 microns in diameter and bacteria are about .1-10 microns depending on the species. Epithelial cells may measure 20-50 microns in diameter depending on the location.
For gross dissection a stereomicroscope is used to carefully identify and ink margins. The total magnification here is equivalent to the low mag of a compound microscope.
Also used in the laboratory are the fluorescent microscope, electron microscope, and laser scanning confocal microscope depending on the needs (usually research and rarely a clinical specimen). The best resolution is obtained with an electron microscope in tissue sections.
The Cell
In pathology, we are mainly concerned with the alterations that occur in the organization of organs, tissues and cells and occasionally in altered organelles. Cells contain various pigment granules, glycogen, lipids depending on their phenotype. These features may be critical to a diagnosis. Nuclear abnormalities are paramount in cancers and are carefully assessed.
Tissue Types-Epithelium, Blood Elements, Muscle etc.
The skin contains a thick multi-layered stratified squamous epithelium. The term stratified refers to the architectural feature that cells are stacked upon one another and joined by intercellular connections. In the skin, stratified squamous epithelium cells at the surface flatten and contain thick proteins named keratins. Therefore, skin is a keratinized stratified squamous epithelium. The skin varies in thickness and in organization all over the body. The skin of the eyelid in thin, that of the hand is thick. Below is a photomicrograph of skin adjacent to the eyelid. Illustrated are general features of skin that will be helpful to learn. Here we will concentrate more on form than function because you are learning what these structures look like under the microscope. The epidermis is the surface lining in this case the stratified squamous epithelium. The dermis lies beneath the epidermis and contains adnexal structures (hair follicles, sebaceous glands, and apocrine glands as well as nerves, blood vessels and soft tissue. The dermis can also be subclassified into the papillary dermis, which is immediately under the epidermis and the reticular dermis, which is deeper and conterminous with underlying subcutaneous fat. The collagen bundles in the papillary dermis are generally thin. The bundles are generally thicker in the reticular dermis. The epidermis is further divided into layers illustrated at relatively high magnification below.
The epidermis is the surface lining in this case the stratified squamous epithelium. The dermis lies beneath the epidermis and contains adnexal structures (hair follicles, sebaceous glands, and apocrine glands as well as nerves, blood vessels and soft tissue. The dermis can also be subclassified into the papillary dermis, which is immediately under the epidermis and the reticular dermis, which is deeper and conterminous with underlying subcutaneous fat. The collagen bundles in the papillary dermis are generally thin. The bundles are generally thicker in the reticular dermis. The epidermis is further divided into layers illustrated at relatively high magnification below. The basal layer is the deepest layer of the epidermis and is composed of a single row of nearly columnar cells with the long axis in line with a perpendicular dropped from the surface of the skin. The stratum spinosum is composed of more polygonally shaped cells with prominent intercellular bridges or spines. As the cells of the stratum spinosum approach the surface they become more flattened. The stratum granulosum is the granular layer characterized by blue keratohyaline granules, the precursor of dense keratin that will form the surface of the epidermis. This surface layer is formed of non-nucleated cells or otherwise known as anucleate squamous cells. There are dendritic cells in the epidermis. Melanocytes may have clearing of the cytoplasm and are generally located in the basal layer. There are occasional Langerhans cells and other dendritic cells. These cells become quite evident in some disorders when they are pigmented. The basal layer of the epidermis is considered the germinative epithelium and gives rise to the more superficial layers that eventually desquamate.
The basal layer is the deepest layer of the epidermis and is composed of a single row of nearly columnar cells with the long axis in line with a perpendicular dropped from the surface of the skin. The stratum spinosum is composed of more polygonally shaped cells with prominent intercellular bridges or spines. As the cells of the stratum spinosum approach the surface they become more flattened. The stratum granulosum is the granular layer characterized by blue keratohyaline granules, the precursor of dense keratin that will form the surface of the epidermis. This surface layer is formed of non-nucleated cells or otherwise known as anucleate squamous cells. There are dendritic cells in the epidermis. Melanocytes may have clearing of the cytoplasm and are generally located in the basal layer. There are occasional Langerhans cells and other dendritic cells. These cells become quite evident in some disorders when they are pigmented. The basal layer of the epidermis is considered the germinative epithelium and gives rise to the more superficial layers that eventually desquamate.
The dermis contains a number of appendages (called epidermal appendages because these all join the epidermis). There are sebaceous glands, eccrine glands and apocrine glands. Below one can see a sebaceous gland that is merging at the follicular infundibulum. 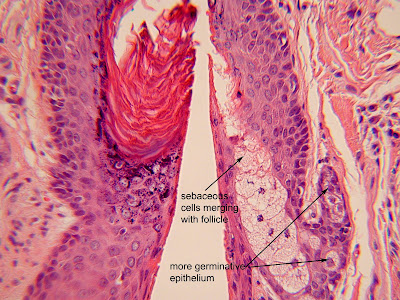 The sebaceous cells at the surface have apoptotic nuclei and those deeper are more intact. There are some cells that are earlier in the transition (toward the germinative condition) and have large nuclear to cytoplasmic ratios and round nuclei with nucleoli. Sebaceous glands are holocrine glands and their secretion includes a variety of cellular components including lipids. Eccrine glands and ducts are present in the dermis as well. Below find both the eccrine glands and the ducts. The myoepithelial cells of eccrine glands are said to be confined at the level of the secretory coil; myoepithelial cells are not found in the long terminal duct that makes its way to surface forming the acrosyringium. Myoepithelial cells are flattened cells that react with smooth muscle antibodies and are found only deep in the secretory coil in the lobules. The eccrine glands are true sweat glands.
The sebaceous cells at the surface have apoptotic nuclei and those deeper are more intact. There are some cells that are earlier in the transition (toward the germinative condition) and have large nuclear to cytoplasmic ratios and round nuclei with nucleoli. Sebaceous glands are holocrine glands and their secretion includes a variety of cellular components including lipids. Eccrine glands and ducts are present in the dermis as well. Below find both the eccrine glands and the ducts. The myoepithelial cells of eccrine glands are said to be confined at the level of the secretory coil; myoepithelial cells are not found in the long terminal duct that makes its way to surface forming the acrosyringium. Myoepithelial cells are flattened cells that react with smooth muscle antibodies and are found only deep in the secretory coil in the lobules. The eccrine glands are true sweat glands.
Apocrine glands are characterized by budding off of the superficial apical epithelium. Below are apocrine glands and their corresponding thin walled ducts that lead eventually to the surface of the skin.  Note the apical pinched off cytoplasm at the tip of the arrow in the apocrine gland. Apocrine are modified sweat glands. The glands are generally of greater diameter than eccrine glands and in contradistinction to eccrine glands, apocrine glands generally show secretory material in the lumen. This distinction has signficance in the discussion of hidrocystomas of eccrine or apocrine origin. Hair shafts arise from the dermis and contain hair papilla, medulla, matrix, inner and outer root sheaths and associated erector muscle. As shown the outer root sheath is composed of cells with a columnar shape and clear cytoplasm that palisade externally. The inner root sheath contains the lone cell layer of Henle that is barely visible at the arrow. The layer of Huxley shows flattened cells with characteristic bright red cytoplasm. More internally located is the matrix of the hair follicle and at the center of the bulbous tip are melanocytes and germinal undifferentiated cells that make up the papilla.
Note the apical pinched off cytoplasm at the tip of the arrow in the apocrine gland. Apocrine are modified sweat glands. The glands are generally of greater diameter than eccrine glands and in contradistinction to eccrine glands, apocrine glands generally show secretory material in the lumen. This distinction has signficance in the discussion of hidrocystomas of eccrine or apocrine origin. Hair shafts arise from the dermis and contain hair papilla, medulla, matrix, inner and outer root sheaths and associated erector muscle. As shown the outer root sheath is composed of cells with a columnar shape and clear cytoplasm that palisade externally. The inner root sheath contains the lone cell layer of Henle that is barely visible at the arrow. The layer of Huxley shows flattened cells with characteristic bright red cytoplasm. More internally located is the matrix of the hair follicle and at the center of the bulbous tip are melanocytes and germinal undifferentiated cells that make up the papilla.  The dermis also contains collagen, blood vessels, muscle, nerves and fat (in the deeper portions).
The dermis also contains collagen, blood vessels, muscle, nerves and fat (in the deeper portions).
The cornea is lined by a non-keratinized stratifed squamous epithelium that is about 50 microns in thickness. The surface two cell layers are called superficial cells. These cells are long (45 microns) and thin (4 microns). The surface of these cells is specialized with microplicae and microvillae that are about .5 to 1 micron in thickness and usually difficult to resolve in routine paraffin embedded specimens. Beneath the superficial cells are 2-5 layers of cells called wing cells that are polygonal in shape, closely interlocked by desmosomes or tight junctions, convex in shape anteriorly and concave posteriorly, and not as long (15-20) microns as superficial cells but are thicker- about 10 microns. Beneath the wing cells reside a layer of cuboidal shaped basal cells that are tall about 18 -20 microns but not long- about 10 microns. The nucleus of the basal cell is round and about 6 microns in diameter. Nuclei of the various layers flatten as one progresses toward the surface to superficial cells. (For images of the cornea please see this link.)
The conjunctiva is generally lined by a specialized non-keratinized stratified squamous epithelium which varies according to its anatomic location, bulbar, forniceal, tarsal, or marginal. Please see this link for details. The bulbar conjunctiva is so christened because it resides on the surface of the eyeball. In the photo of conjunctiva below there is inflammation leading to some cell pathology but the general architecture is discernible with stratified squamous epithelium. At the base, the basal cells have a more cuboidal shape. Toward the surface the cells begin to flatten (arrow 1). The junctions between cells are faintly visible in the layers near arrow 2 as tiny thin vertical lines between 2 cells. These are tight junctions known as desmosomes but under the light microscope are called intercellular bridges. Can you name the other cells at the arrows? The answers are provided below.

The lacrimal gland is composed of roughly circular arrangements of cuboidal to columnar shaped cells. Each of these circular structures is called an acinus. The acinus secretes the fluid of the lacrimal gland. Each acinus contains contractile myoepithelial cells (spindle shaped cells) in the outer aspect. The acini are organized into lobules that drain into a ductular system. The intralobular ductules connect to the acini, which join the interlobular ducts and eventually converge into the main excretory ducts of the lacrimal gland. Lacrimal epithelial cells (or acinar cells) have a specific orientation with the nuclei oriented toward the basal surface and the zymogen granules (proteins) oriented toward the lumen. Ductules lack the spindle shaped myoepithelial cells that surround the acini. (See link for photos of the lacrimal gland and more detail.)
2. Blood- Blood contains red blood cells, white blood cells, platelets, and plasma. The red blood cells are also called erythrocytes and are membrane bags that contain hemoglobin to transport oxygen. The red blood cell is biconcave and has a disc shape. Mature red blood cells have no nucleus. The diameter of the erythrocyte is about 7 microns. The photo above of the conjunctiva shows erythrocytes at arrow 3. You can even see the biconcave disc in arrow 3 that veers to the right.
White blood cells or leukocytes are involved in host defense and mediate inflammatory processes. White blood cells include neutrophils or polymorphonuclear leukocytes, monocytes, lymphocytes, eosinophils and basophils. Neutrophils make up about 50-80% of the total white blood cells. The diameter of the neutrophil is about 10-12 microns. The cells is round but has a distinctive nucleus that is multi-lobated. The cytoplasm contains granules that are azurophilic (primary) and other secondary granules that lack enzymes. Neutrophils derive their name from the secondary granules because they stain with neutral dyes. Neutrophils are phagocytic for bacteria and increase markedly in tissue that are infected with bacteria. The neutrophil is shown in the photo above at arrow 4. There are others in the photo but this is unmistakable for multiple nuclear lobes joined by narrow segments. Lymphocytes compose about 25% of the white blood cells, are spherical, and measure about 8-12 microns in diameter. The lymphocyte has scant cytoplasm and a round nucleus. The nucleus has dark areas with chromatin that form a peripheral rim with clumps located more centrally. There are light areas in between. The nucleus is about the size of an erythrocytes. The lymphocyte is critical to immunological defense for the body. Lymphocytes are seen in tissue during long standing or chronic inflammation. Arrow 5 in the photo above shows a lymphocyte with a round nucleus and vary scant cytoplasm. Note the size of the nucleus approaches that of the neighboring red blood cells at arrow 3.
Eosinophils are leukocytes that account for only 1-2% of white blood cells. The average size is 10-15 microns and they have a characteristic bilobed nucleus that lies peripherally in the cell and very large red cytoplasmic granules when stained with eosin (hence the name). The granules are about 1 micron in diameter and are resolvable under the light microscope. An eosinophil is illustrated in the high power image below at arrow 6. One can make out the a few individual granules both intracytoplasmic although at this magnification challenges the limits of resolution of the microscope.

Monocytes comprise about 5% of leukocytes in the blood. The monocyte diameter is about 15 microns and a characteristic kidney or bean shaped nucleus lies eccentrically in the cells. The monocyte cytoplasm is abundant and vacuolated. Monocytes are the precursor to tissue cells called macrophages that are powerful phagocytic cells. Macrophages are a marker for a longstanding inflammatory process or infection. A tissue macrophage is evident at arrow 7 in the image above. The green in the photo above is marking ink used to identify the margin of the specimen. The section was cut tangentially so some of the margin appears in the center of the specimen.
Plasma cells are mature immune cells that produce antibodies or immunoglobulin. These cells are derived from lymphocytes of B cell lineage. The plasma cell varies in size from about 10 microns to 15 microns and occasionally higher with very reactive cells (see arrow 8). The nucleus of the plasma cell is distinctive, featuring a dense dark chromatin segments arranged at the periphery of the nucleus. This appearance is coined a "clock face" (arrow 9). The nucleus is consistently seen at the periphery or eccentrically in the cell. The classic plasma cell has a prominent clear area in the cytoplasm corresponding to the Golgi apparatus (arrow 10). The plasma cell is replete with machinery to produce protein (antibodies) so it contains abundant rough endoplasmic reticulum. The cytoplasm has a distinctive purple appearance, other than the  perinuclear clearing of the Golgi.
perinuclear clearing of the Golgi.
3. Muscle There are 3 basic types of muscle. Two types. striated and smooth, concern ocular pathology specimens. Striated muscle has unique characteristics that aid in recognition. Cross striations are seen in longitudnal sections of the muscle. The nuclei are peripheral in the cell in both longitudnal sections and cross sections. A typical example of striated muscle in the ocular adnexa (structures surrounding the eye) is the ocularis oculi. This is a muscle that helps control the closure of the eyelid. It has been dubbed the sphincter of the eye! The image below shows the peripheral nucleus and cross striations of peripheral muscle. The orbicularis oculi has connections to the eyebrow and the cheek.  Extraocular muscle are striated muscles that are renowned for the highest level of innervation in the body. The image below is a histologic photograph of an extraocular muscle that is stained with a trichrome stain. The trichrome stain dyes muscle red and fibrous tissue blue.. Note the numerous cross striations (arrow) of the red muscle and then the dense fibrous tissue which composes the tendon (blue).
Extraocular muscle are striated muscles that are renowned for the highest level of innervation in the body. The image below is a histologic photograph of an extraocular muscle that is stained with a trichrome stain. The trichrome stain dyes muscle red and fibrous tissue blue.. Note the numerous cross striations (arrow) of the red muscle and then the dense fibrous tissue which composes the tendon (blue). 
4. Nerves Peripheral nerves, as opposed to nerves of the central nervous system (brain) are easy to recognize because they are myelinated. The nerves are surrounded by a fibrous sheath called an perineurium and contain axons that are small pink structures surrounded by myelin composed of lipids that appear clear in histologic section. The image below is a nerve with all of the key features labeled.  Nerves are important in eye pathology because tumors may arise from the nerve sheath.
Nerves are important in eye pathology because tumors may arise from the nerve sheath.
5. Blood vessels. Blood vessels supply blood and nutrients to tissues throughout the body. The major categories include arteries, veins and capillaries. Arteries are thick walled conduits that receive blood from the heart under fairly high pressure. Arteries are round structures when seen in cross section and have lumens filled with erythrocytes (arrow 12). The outside of the artery is covered with a fibrous adventitial sheath (arrow 13). The arterial wall contains smooth muscle cells (arrow 14) that are spindle in shape and contain myofibrils. Arteries have elastic tissue that allow expansion under pressure called an internal elastic lamina (arrow 15). The arterial lumen is lined by single cells called endothelium (arrow 16). 
Capillaries are thin walled often lined by a simple single layer of endothelium. Diffusion occurs through the thinned walled vessels to deliver nutrients to tissues. Veins, return blood to the heart and have some smooth muscle and elastic tissue but much less prominent in than arteries. Veins have thinner walls than do arteries.
